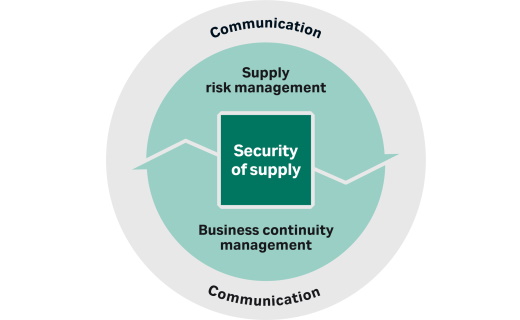
-
Quality by design
System suitability, usability and compliance starts from the moment we start the design process.
-
Single-use formulation and filling
A case study.
-
ADC extractables studies
Performed with two solvents commonly used in the ADC cytotoxin conjugation step: DMA and DMSO.
-
Single-use and sustainability
Although it might seem counterintuitive, they go hand in hand.
-
Allegro™ Ready single-use systems
Standard designs and short lead times, ready for when you need them.
-
Flowstar LGR test instrument
In-situ leak testing for quality risk management.
-
Validation services
Technical, regulatory, and scientific consultancy to support process qualification and validation.
-
Batch 360™ single-use consumables planning
Design and visualize your single-use process to generate a detailed bill of materials, and so much more.
-
Related solutions
- Economic simulation
- Regulatory documents
-
Enabling services
- Manufacturing
- Custom
- In-person training
- Process development