Blog
Pharmaceutical Production Platforms – and other P’s
August 3, 2022
Pharmaceutical Production Platforms – and other Ps
So what is a pharmaceutical production platform? In its most simple form, a pharmaceutical platform process is a standardized collection of preselected technologies that combine to support the manufacture of more than one drug product. When well designed, it fast tracks the creation of a process using proven, well characterized technology and performs at a level that is close to the level that may be expected from alternative technologies or products. But when the limits of the platform are exceeded, it artificially restrains the productivity when compared to the same reference point and may demand compromises in terms of cost, achievable processing speed, capacity or any other measure, and that typically has a detrimental impact upon productivity.
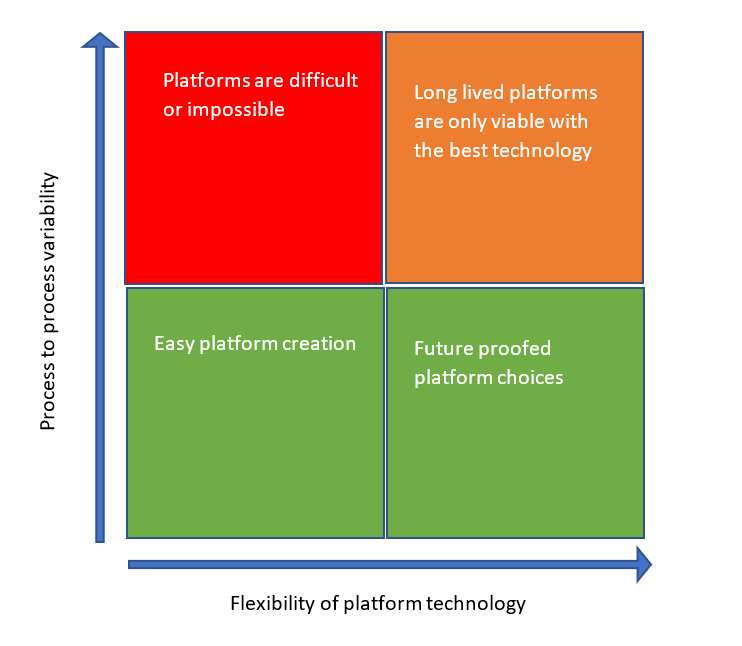
The production platform is the ultimate expression of confidence in the ability of a manufacturing solution to be able to adapt to the production of multiple, similar products. The trust in, and viability of, any platform depends on two main variables. How different these similar products actually are, and how flexible the technology choices within the platform are to cope with changing production challenges. For many processes there is a large overlap with similar products providing very similar challenges to the chosen technology within the platform but, the further forwards we look, the greater the likely need for change. Only flexible platforms with the features and performance characteristics that can cope with change can fully support this.
Potential for change is something that is often difficult to plan for. While change does not normally happen quickly, which would demand significant foresight for continued success, it can happen. This was proven by the rapid rise in viral vector and mRNA COVID-19 vaccines and the global scramble for CDMO capacity and capability. Many manufacturing facilities specialize in the production of a specific drug type, such as monoclonal antibodies, and to a lesser extent viral vectors, and fortunately the manufacturing platforms for these vaccines were not dissimilar to those for other products. Platforms, both new and old played a significant role in supporting the accelerated creation of these processes at the scales demanded by the pandemic. Change can also occur organically as process intensification within an otherwise unchanged platform alters the demands on one or more unit operation. Change is usually a good thing, albeit something that is often resisted due to the amount of paperwork it can generate. Change therefore does not typically happen without good reason or without a clear and significant driver for that change.
Performance, or more accurately the robustness and versatility of the performance of any technology within the platform, has a direct impact upon the longevity of any platform in the presence of any shift in different process needs over time. Any technology that operates efficiently within a broad design space has a distinct advantage over those that may already be operating at the limits of one of its performance characteristics. The best technologies and the best platforms accommodate change and help the process evolve as new challenges are overcome and new opportunities are grasped. Without the opportunity for first-hand development through experience and trial (and invariably error), designing a platform often benefits from external guidance. When identified, the best platform solution fast tracks and safeguards the choices that are right for the process and business needs, both in the short and medium term.
Practicality and usability of any platform is a major factor in its success and over time can be complicated by the periodic substitution of individual technologies when these fail to meet the needs of the process or business. Change can be painful, and the idealized vision of a perfect platform process is one that is able to utilize the best technology at the unit operation level and to bring these unit operations together seamlessly in a coordinated and fully automated process that can embrace change. Achieving this requires process elements to be flexible and to be configured in such a way as to work with adjacent unit operations both now and with whatever comes next.
Without a degree of flexibility the benefits of any platform technology can be restricted and the platform may be destined to have a short relevance. Rigid inflexible platforms limit the ability of the platform to evolve to meet changing needs. Platform technology that can quickly adapt to different needs, however, can support a wide range of processes and encourages evolution to realize improvements in productivity and efficiency.
To learn more about Pall’s platform solutions visit our process workflow collection.

Mark Ayles, Senior Marketing Manager
- Category
- Author
- Sort By