In general, you should select a MWCO that is 3 to 6 times smaller than the molecular weight of the protein or other species to be retained. If flow rate or processing time is a main consideration, choose a membrane with a MWCO at the lower end of this range (3x); if retention is the main objective, choose a tighter membrane (6x). These values should be considered a guide, as solute retention and selectivity can vary depending on many factors such as pressure, molecular shape, presence of other solutes, and ionic conditions. Due to their complex shape, proteins behave differently than nucleic acids, which are linear and flexible. For the latter, suitability of centrifugal device MWCO is correlated with fragment length range. Because linear nucleic acid molecules can be forced through many MWCO membranes regardless of size, effective retention by an ultrafiltration membrane will require a reduction in g-force. For maximal nucleic acid retention, Nanosep device speeds must not exceed 5,000 x g and for the other centrifugal devices a speed at the lower end of the recommended g-force range is recommended.
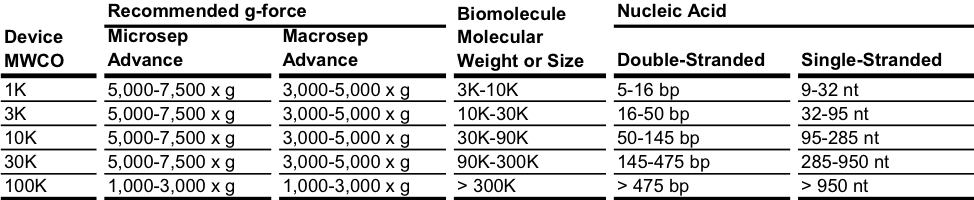
The 100K membrane has 10 nm nominal pore size and is suitable for Biomolecule/particles larger than 30 nm. Centrifugation with nucleic acids is recommended to be carried out centrifugation at the lower end of centrifugal force range
If using a fixed angle rotor, the membrane paddle in the sample reservoir of the centrifugal device should be on the radial axis of the rotor as indicated in the drawing below. Although incorrect orientation has no effect on membrane integrity or retention, it will increase the processing time and impact the final achievable sample concentration factor.
Pall offers four different centrifugal devices meant for processing samples of increasing volume. The Nanosep® device is intended for filtering samples with volumes of up to 500 µL; the Microsep™ Advance device is for samples up to 5 mL; the Macrosep® Advance device for samples up to 20 mL; and the Jumbosep™ device is for samples up to 60 mL. The Jumbosep device's housing is reusable and you only have to replace the membrane inserts between experiments. The other three devices are meant for single use only.
To find these products please visit Centrifugal Devices
No, the devices are non-sterile. The devices can be sanitized by filtering 70% ethanol through them prior to use.
Certain proteins, particularly when dilute, tend to show a high affinity towards various surfaces which may lead to irreversible binding. Although Pall centrifugal devices are made of materials with low intrinsic affinity for proteins, this phenomenon can cause decreased recovery. Protein recovery can be improved through 'passivation' of the device. This is a pretreatment aimed to occupy potential binding sites by filtering a solution with additional protein (often albumin), detergents, or salts through the centrifugal device prior to use.
The sample occupies a contiguous channel perpendicular to the filter membrane paddle at the bottom of the sample reservoir and therefore it is not necessary to aspirate the retentate from both sides of the filter membrane paddle. Simply place the pipettor into the channel and draw out the retentate.
Sodium azide is added to the Omega™ modified polyethersulfone ultrafiltration membrane as a bacteriostatic preservative. As sodium azide inhibits cytochrome oxidase in the mitochondrial electron transport chain and can induce apoptosis, its presence in filtrates may have undesired effects on living cells or in in vivo studies. Sodium azide can also interfere with amine group-dependent conjugation reactions. If these or similar kind of problems are anticipated, a pre-rinse step can be performed to remove sodium azide from the membrane prior to filtering samples. Please note that the device should be used within 20 minutes of completing the pre-rinse step to prevent irreversible membrane damage due to dehydration.
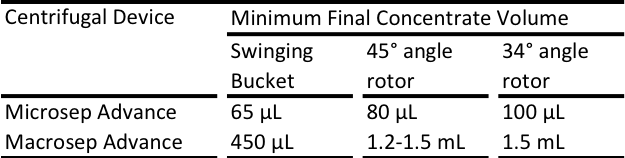
The total volume of liquid in the Microsep Advance and Nanosep centrifugal devices determines the final retentate volume. By adding buffer under the device insert, you can set your dead stop volume and thereby select the concentration factor. This link will allow you to download the Concentration Selection Guide for Nanosep and Microsep Advance Centrifugal Devices which contains all the information on how to do this.
The colors reflect the MWCO of the Omega membrane
Yellow – 1K
Gray - 3K
Blue - 10K
Red - 30K
Clear - 100K
Orange - 300K
The pre-rinsing step to remove the trace amounts of glycerin and sodium azide present in the Omega™ modified polyethersulfone ultrafiltration membrane and sanitize the devices should be performed prior to sanitizing after the centrifugal device.
UV treatment of Microsep and Macrosep devices has not been tested. Membrane degradation and loss of integrity could be of concern. Sanitization by filtering 70% ethanol through the devices prior to use is recommended.
The retentate should be easily recoverable by tilting the sample reservoir to the side, and pipetting the liquid out (may need a 1mL pipette tip to recover from the Macrosep® Advance Centrifugal Devices). Removing the sample reservoir from the main tube should help make it easier to see/remove the sample.
The Microsep and Macrosep devices have not been tested for compatibility with gamma irradiation. The devices can be sanitized by filtering 70% ethanol through them prior to use.
Pall has a mature Business Continuity Management System, as well as a strong global and local infrastructure, to ensure minimal disruption to our business. In addition, Pall has implemented a comprehensive COVID-19 Pandemic Response Plan to minimize any impact to our customers, while protecting our associates and communities.
Pall prioritizes our associates’ health and well-being, as they safely work to manufacture and deliver essential products and services to customers. We take measures to prevent the spread of COVID-19 at our sites and promote social distancing.
Pall solutions play a significant role in the fight against COVID-19, from breathing filters for ventilators, to HEPA filters for aircraft cabin air quality and vaccine scale up manufacturing processes. Pall solutions are also used around the globe in COVID-19 research.
The AcroPrep 24-well filter plate can be used in centrifuges equipped with a compatible swinging‑bucket rotor. The recommended centrifugal force is 1,000 × g.
Important Note: The filter plate comes with its dedicated receiver plate and when stacked together they have a combined height of 7.5 cm (2.97 in.). For trouble-free operation with swinging-bucket rotors in the centrifuge, it is essential to verify that there is enough rotor clearance to allow uninhibited swinging motion of the microplate carrier with the filter plate stack.
The AcroPrep 24-well filter plate has been designed to comply with the ANSI/SLAS X-2004 microplate standards, which means that it has a footprint with a length of 12.8 cm (5.0 in.) and a width of 8.6 cm (3.4 in.). The filter plate has a height of 3.9 cm (1.5 in.). The filter plate comes with its dedicated receiver plate which has the same dimensions. When the receiver plate and filter plate are stacked, they have a combined height of 7.5 cm (2.97 in.).
Note: The latter stack height is an important consideration for use with swinging-bucket rotors in the centrifuge. It is essential to verify that there is enough rotor clearance to allow uninhibited swinging motion of the microplate carriers with the filter plate stack to enable trouble-free centrifuge operation.
Yes, both the AcroPrep 24-well cell clarification and sterile filtration plate and the AcroPrep 24-well sterile filtration plate are individually packaged gamma-irradiated devices that provide sterile grade filtration. The plates provide a sterile effluent following liquid challenge with Brevundimonas diminuta under laboratory conditions at a challenge level of ≥107 cfu/cm².The AcroPrep 24-well cell clarification and sterile filtration plate combines cell clarification and sterile filtration in a single workflow step.
The AcroPrep 24-well filter plate can be used for vacuum processing. The Pall multi-well plate vacuum manifold (part number 5017) is well suited for this application. The recommended operating vacuum is ≥ 25.4 cmHg (10 inHg). In experiments with cultured mammalian cells (CHO and HEK293T), a vacuum of 38.1 cmHg (15 inHg) was found to work well.
The hold-up volume of a filter plate is the volume of sample that is retained by the plate and cannot be recovered following filtration. The typical hold-up volume for the AcroPrep 24-well cell clarification and sterile filtration plate is specified as < 0.4 mL/well.
During development, the AcroPrep 24-well cell clarification and sterile filtration plate has been used successfully for clarification of 5 mL cell cultures with densities of > 25 x 106 cells / mL at 90% viability (CHO and HEK293T).
The AcroPrep 24-well filter plate has a 7 mL well volume. All this capacity is available for processing via vacuum, where the recommended working volume is 7 mL. However, for centrifugation, the use of a swinging-bucket rotor limits the recommended working volume to 6 mL.
The hold-up volume of a filter plate is the volume of sample that is retained by the plate and cannot be recovered following filtration. The typical hold-up volume for the AcroPrep 24-well sterile filtration plate is specified as < 0.2 mL/well.
Pall is participating in a consortium led by the Jenner Institute, University of Oxford (Oxford, England), to rapidly develop, scale-up and produce a vaccine for COVID-19. As part of the consortium, Pall will develop a large-scale manufacturing process, so that millions of doses of a vaccine can be produced. You can learn more here: Pall Corporation Participates In A Consortium To Rapidly Develop A COVID-19 Vaccine.
The filters in each well of the filter plate have an EFA of 1.57 cm² (0.243 in²).
Nanosep® centrifugal devices do not have a dead stop volume, but some moisture is always retained in the membrane. Therefore, even if the device appears to have spun to dryness, in most cases the retentate can be recovered by pipetting a small volume of buffer over the surface of the membrane. It is how ever possible to set a dead stop volume by placing buffer below the insert, please refer to the Nanosep® concentration guide. The Microsep™ Advance and Macrosep® Advance Centrifugal Devices do have built-in dead stop volumes.
No, cross-talk is not a problem with the AcroPrep 24-well filter plates when samples are processed as recommended.
With filter plates, the term cross-talk is used when filtrate from a given well is found in adjacent wells. In principle cross-talk can occur prior to filtration by weeping of sample from one filter plate well to another or during filtration by deposition of the filtrate in adjacent wells of the receiver plate.
As the sample wells of the AcroPrep 24-well filter plates are completely independent, there is no possibility of a sample finding its way from one well to another prior to filtration. This, combined with the larger outlet tip-to-tip distances of the 24-well filter plate compared to those found in traditional 96-well or 384-well filter plates, means that cross-talk should not be a concern if the maximum processing volumes are adhered to.
Pall’s BOS filter bags are constructed entirely without seams. The unique material composition of the Polymicro® seamless filter bags provides high efficiency with graded pore-size distribution and facilitates absolute filtration.
The Forward Flow Test is designed to test the integrity of sterilizing and virus grade hydrophilic and hydrophobic membrane filters. This qualitative test is based on measuring the gas flow across a completely wetted membrane at a defined constant test pressure on the upstream side. When the downstream side of the membrane is at atmospheric pressure a diffusion flow of gas is established due to the pressure differential. The Forward Flow test is Pall’s recommended integrity test for capsule and cartridge filters.
Yes, the SUTFF module requires a SUTFF module holder. The SUTFF module must be placed in the SUTFF module holder and compressed to activate the seals. The SUTFF module will leak if it is used without a module holder. Refer to FAQ: What is the part number of the SUTFF module holder?
Measured water flow that exceeds the test limit during Water Intrusion testing can be caused by a range of root causes, some of which are listed below. It can be seen that there are several factors which can lead to false test failures.
Possible root causes for false failures are:
- System leaks (filter housing, fittings, tubes, etc.)
- Insufficient test time
- Insufficient stabilization time
- Temperature influence
- Reversible partial wetting of the filter membrane (due to condensation of moisture within the pores or excessive pressure events)
More unlikely for unused filters (pre use test):
- Foreign substances / contaminations deposited on the filter
Root causes for true failures are:
- Filter defects
- Compromised O-ring seal with the housing
The WIT is performed on a non-wetted (“dry”) hydrophobic filter. The upstream side of the filter assembly is completely filled with water, covering the entire filter. An air test pressure lower than the actual water intrusion (or water breakthrough) pressure of the largest membranes pores is applied to the system. The membrane pores remain “dry” during the test. The test pressure drives a transport of water vapors from the water phase across the filter membrane following the pressure differential. Transport of liquid water through all wetted pores and wetted flow pathways or defects will also occur. The WIT quantitatively measures the sum of water vapor (evaporation) and liquid water flow through the hydrophobic filter.
For a sterilizing grade filter, the maximum allowed flow (integrity test limit) is derived from the generic filter validation and correlated with the bacteria retention capability of the filter.
For more information, please see Pall Publication, USD 3033 Application Note: Best Practices for Successful Filter Integrity Testing Using the Water Intrusion Test (WIT) Method
Pall recommends the use of water to lubricate the O-rings of a filter in order to ease installation into the housing.
Pall does not recommend the use of alcohol (or an alcohol/water mixture) as it could come into contact with the filter membrane, causing a hydrophilic spot that will allow water passage, resulting in a false failure test result, especially in a water intrusion test.
Additionally, if any alcohol gets trapped between the two O-rings grooves, there will be a localized area where the alcohol will expand when the filter is sterilized (autoclave or steam-in-place). This can potentially damage the filter adapter, or impact the filter to housing seal.
For more information, please see Pall Publication, USD 3033 Application Note: Best Practices for Successful Filter Integrity Testing Using the Water Intrusion Test (WIT) Method
In general, Pall does not recommend performing an integrity test at temperatures above 50°C when using water as the wetting fluid.
Forward Flow measurements at temperatures above 50 °C are considerably higher than at ambient temperature, and are more difficult to keep constant to the ±1 °C test specification. This may introduce inaccuracies in the measurement and in calculating appropriate test limits. When integrity testing is performed at elevated temperatures, the use of jacketed housing or a heating jacket is recommended to keep the temperature stable during the test.
While Pall can provide calculated test limits for elevated temperatures when water is used as the wetting fluid, additional testing should be performed to confirm these limits. In addition, monitoring actual test results by the end user can also show that these limits are appropriate.
Please contact a Pall representative if you would like more information on developing Forward Flow and Bubble Point limits for all wetting fluids at elevated temperatures.
The Bubble Point Test is designed to detect the largest pores of hydrophilic and hydrophobic membrane filters. The Bubble Point test is based on measuring the gas flow across a completely wetted membrane at increasing gas test pressure, until the point at which the wetting fluid is expelled from the pores, and bulk flow is measured.
The Bubble Point test is considered a subjective test, and the results can vary depending on the algorithm of the test instrument. The Bubble Point method is the preferred integrity test method for testing filter discs, as the Forward Flow is often too low to be accurately measured.
The Water Intrusion Test measures water flow through a submerged filter when pressure is applied to the upstream side of the filter housing. Because this test can only be performed on a hydrophobic filter, the WIT measurement for an integral filter is primarily evaporative flow of water through the pores of the membrane.
When a hydrophobic filter becomes partially wetted with a low surface tension liquid such as an alcohol water mix, or condensate (from autoclave or Steam-In-Place (SIP)), then the WIT may result in a false failure. This is due to a water channel forming through the membrane in areas where it has become wet, resulting in the free flow of water.
If the filter has become partially wet, it must be restored to a fully dry state before a WIT can be performed successfully. Flowing compressed air through the filter for several hours is often required. Alternatively, oven drying can be performed. Please contact your local Pall representative for the appropriate filter drying conditions.
Pall recommends the following to prevent a hydrophobic filter from becoming wet:
- Keep the filter away from potential sources of low surface tension liquids such as alcohol mixtures.
- If the filter is autoclaved, use a slow exhaust cycle and a vacuum drying cycle.
- If the filter is subjected to SIP, use a cooling gas such as air or nitrogen following SIP.
In certain applications, post-use testing using WIT is impractical due to product contamination on the filter, i.e. bioreactor exhaust filter. In these cases, Pall recommends performing a post-use Forward Flow integrity test.
For more information, please see Pall Publications:
The Water Intrusion Test is a practical and validated test which can be used for in-situ integrity testing of hydrophobic gas filters. This test is conducted with deionized or higher quality water, without the need for low-surface-tension flammable solvents (such as Isopropyl Alcohol or Ethanol). Due to occupational risks, environmental regulations, safety guidelines and cost associated with handling these low surface tension solvents, water intrusion is becoming the method of choice for integrity testing hydrophobic microbial rated filters for air or gas applications.
Water Intrusion testing is the preferred test where the hydrophobic microbial rated gas filter:
- Is integrity tested in-situ
- A pre-use test is performed (especially after sterilization)
- Alcohol use is restricted or not allowed in the production area
The Forward Flow or Bubble Point test, is the preferred method to integrity test filters in applications where:
- A small area filter is used
- An off-line filter integrity test is performed
- To confirm filter integrity following a water intrusion test failure evaluation
For more information, please see Pall Publication, USD 3033 Application Note: Best Practices for Successful Filter Integrity Testing Using the Water Intrusion Test (WIT) Method
To successfully perform a Water Intrusion Test (WIT), the entire length of the filter has to be covered by (submerged in) water for the duration of the test.
During pressurization of the filter housing assembly to the water intrusion test pressure, the water level drops upstream of the filter. The reason is due to compression of the membrane pleats and elimination of gas bubbles during initial pressurization.
This can leave a portion of the filter exposed to pressurized air, which will freely flow through the exposed area and result in a false test failure.
To troubleshoot the cause of a test failure, Pall recommends:
· To refill the filter housing with water and repeat the test, or
· Before the test, increase the upstream volume, to have more water above the filter, to compensate for the reduced water levels resulting from the compression.
For more information, please see Pall Publication, USD 3033 Application Note: Best Practices for Successful Filter Integrity Testing Using the Water Intrusion Test (WIT) Method
There is no general rule to this and depends on the filter application.
The user should qualify the filter change-out frequency for their specific application, but this should never exceed Pall claims for cumulative sterilization cycles and/or the maximum differential pressure. These specificationswere established by Pall in controlled laboratory conditions with filters that were not exposed to any process (including manufacturing or production) conditions.
The filter lifetime and change-out should be based on a risk assessment by the user for their specific application including relevant validation/qualification data.
For example, the impact of a post-use integrity test failure of the filter should be considered.
For gas filtration in compressed gas, tank vent, or utilities setting, industry best practice is to replace the filter under a pre-defined preventative maintenance schedule (i.e. at a minimum of 12-month cycle).
For critical applications, single use is recommended to eliminate the risks of cross contamination between batches.
For more information, please see Pall Publication:
For gas filtration in a vent application, YES, Pall sterilizing grade gas filters can be used in both directions.
For gas filtration, Pall offers the following sterilizing grade gas filters:
Emflon® II V002PV
Emflon PFR
Emflon HTPFR
Acro® 50 (6074270)
Acrodisc® KM292HP
These filter devices feature a symmetrical filter media construction, where two layers of membrane of the same pore rating are used in the manufacturing of the final filter cartridge or capsule.
When a filter of a symmetrical media construction is used for venting purposes, in either forward or reverse flow direction, the gas flow and any contaminants will travel through a torturous path of the same characteristic and length as in the forward direction. This will lead to the same retention efficiency in either flow direction.
Based on the symmetric media construction the filter will thus act as a sterile barrier when venting in either flow direction. Therefore, Pall Emflon® II V002PV, Emflon PFR, Emflon HTPFR, Acro® 50 (6074270) and Acrodisc® KM292HP filters can be used for bi-directional flow applications under this venting mode of filtration.
In order to consider the influence of the temperature, each filter integrity test method needs to be treated independently, as they are based on different physical principles.
Forward Flow
Forward Flow integrity test limits issued for Pall filters, when wet with standard wetting fluids such as water, 60/40 IPA or similar solutions, apply to a test temperature of 20 °C ± 5 °C.
Any variation in the temperature of any gas volume in the filter test assembly during the measurement phase has an effect on the flow measurement. Most integrity test instruments measure Forward Flow on the upstream side of filter, as a function of gas pressure change1. Variations in temperature during the test lead to expansion or compression of the gas in the test assembly (tubing and housing upstream of the filter). Such variations of the gas volume may lead to inaccurate flow measurements. Therefore, it is recommended to keep the temperature of the filter assembly constant during the test period.
Pall recommends that the temperature of the filter assembly during the test should not vary more than ± 1 °C. Some simple ways to accomplish this are listed below.
Bubble Point
Changes of surface tension are of direct relevance for bubble point testing as the surface tension influences the capillary forces holding the wetting liquid in the membrane pores. The measured bubble point of a given filter which is fully wetted will increase and decrease in direct proportion with the surface tension of the wetting liquid.
This means that lower bubble point values will be measured at higher temperature, and higher bubble point values will be measured at lower temperature.
Water Intrusion Test (WIT)
For Pall filters, the water intrusion limit values apply to ambient temperature (20 °C) with a specified range of ± 2 °C. During the test period, the temperature of the filter assembly should not vary more than ± 1 °C.
For the WIT, the temperature of the gas in the filter assembly will have the same effects on the gas volume as indicated above for the Forward Flow test. Water temperature will also have an effect on the measurements.
As the WIT measures evaporative flow, which is typically much lower than diffusion measurements (as measured by the Forward Flow test), any temperature changes will have a greater effect on the WIT measurements compared to the Forward Flow test and may not be identified by environmental or assembly temperature monitoring.
Maintaining Constant Temperature During Integrity Testing
The following is a list of approaches to maintaining a constant temperature during integrity testing:
- Acclimate the filter and fluids to room temperature before starting the test. This is especially important for the WIT: Our guidance is to dispense the water into a container and acclimate for >4 hours.
- Avoid placing the filter assembly under heating or cooling registers.
- Avoid handling the filter during the test.
- In cases where room temperature fluctuates, it may be necessary to insulate tubing and filter assembly.
1 The Palltronic Flowstar line of integrity test instruments measure Forward Flow by direct measurement. The impact of a change in temperature during the measurement (“Test”) phase will result in an unstable flow measurement, which will extend the test time.
Pall Corporation’s approach for testing multi-modular, sterilizing-grade filter assemblies is to provide assembly-specific Forward Flow limit values below the sum of the maximum allowable FF limit values for the individual filter modules (i.e. a multiplying or reducing factor). This reduction in allowable Forward Flow is designed to reduce the risk that a marginal filter test failure (in the unlikely event that one is present) cannot be detected in a multi-element assembly. It results in a tighter, more conservative test limit when compared to a linear multiplier.
While the use of multiplying factors is not a regulatory requirement, it is Pall Corporations’ philosophy on integrity testing to use practices that provide the maximum safety for large filter area installations. This needs to be balanced with the risk that a set of integral modules may fail the test due to the application of a multiplying factor that is too stringent (“false fail”). This approach is also described in the 2008 revision of PDA Technical Report No. 26 on Sterilizing Filtration of Liquids 1.
Pall Corporation’s basis for defining the appropriate multiplying factor for a specific multi-modular installation is based on several parameters including:
- Type of membrane and number of elements
- Statistical distribution of observed Forward Flow (FF) values for the specific filter
- Mean deviation for Forward Flow distribution
- Standard deviation of Forward Flow distribution
- Point of first failure during microbial challenge (if observed)
Forward Flow limits obtained from Pall Corporation for multi-element filter assemblies will incorporate the appropriate multiplier.
PDA Journal of Pharmaceutical Science and Technology, Technical Report No. 26 Sterilizing Filtration of Liquids, Rev. 2008. Supplement Volume 62, No. S-5
Prior to performing an integrity test, it is imperative that the pore structure of the filter membrane be filled with the wetting fluid. To assure complete wetting, Pall recommends applying downstream flow restriction (sometimes referred to as back pressure, not to be confused with reverse pressure) during the filter flush.
The use of a downstream flow restriction helps to:
- Ensure uniform flow distribution through the entire length of the filter
- Overcome the tendency for fluid to flow through the path of least resistance.
- Remove air entrapped in the membrane pleats, by further solubilizing the air (due to increased pressure in the system) and by compressing air bubbles to a size where they can freely pass through the membrane.
When testing filters larger than 254 mm (10 in.) or multi-round systems, a downstream flow restriction will ensure the fluid reaches the top of the housing during venting. Otherwise, the hydrostatic pressure of the liquid will cause it to flow out of the downstream side of the housing before reaching the vent at the highest point in the system.
To apply a downstream flow restriction, Pall recommends installing a pressure gauge downstream of the filter housing, followed by a flow control valve such as a diaphragm valve.
For more information, please see Pall Publication, USD3297_User Guide: Wetting and Flushing of Pall Microbially_Rated_Filter Cartridges and Capsules
Pall manufactures different membrane filters for gas filtration. We use either Polytetrafluoroethylene (PTFE) filter membrane or Polyvinylidene Fluoride (PVDF) modified, for such a purpose.
In our Pall Allegro™ single-use systems, the method to sterilize these systems is by gamma irradiation.
The Polytetrafluoroethylene (PTFE) filter membrane, is incompatible with gamma irradiation, so it could not be used as a gas filter in our Single-Use Systems.
The Polyvinylidene Fluoride (PVDF) modified is compatible with gamma irradiation and for this reason is the device to be used with our Single-Use-Systems.
Pall Emflon II V002 sterilizing grade filters uses a filter membrane made of polyvinylidene difluoride (PVDF).
Integrity test failures can be caused by a range of root causes, some of which are listed below:
- System leaks (filter housing, fittings, tubes, etc.)
- Insufficient test time
- Incorrect temperature of wetting fluid
- Insufficient stabilization time
- Temperature influence
- Incomplete wetting of the filter
- Incorrect test limits
- Incorrect pressure source
- Filter defects or damage
- Compromised O-ring seal
- Incorrect filter selection
When an integrity test failure occurs, the first step is to verify the system setup and test parameters. After all of these conditions have been verified, the filter should be re-wet and tested again (using the Forward Flow test, even if the initial failure was recorded using a bubble point test, except for small area filters (<200cm2).
If the result is a pass, then the filter is integral. If it fails, the Forward Flow test should be repeated after a more vigorous wetting step. This can include a larger flush volume, application of back pressure (downstream flow restriction), or increased differential pressure. If the filter fails again, a flush and Forward Flow integrity test with a low surface tension wetting fluid, such as 60:40 IPA/water, should be performed. If the filter fails the integrity test again, the filter (still installed in the housing if possible) should be returned to Pall Corporation for further analysis.
When investigating an integrity test failure, one of the first steps is to determine if any leaks are present in the system under test. Integrity test instruments perform the measurements on the upstream side of the filter, thus preserving the downstream sterility of the system. The test instrument is not able to differentiate gas flow through the filter from gas flow through a leak in the hardware.
Prior to performing the leak test, a visual inspection of the system is recommended to identify any potential leak sources. Damage to the housing or to the o-ring seals are common sources of leaks. Such signs of damage include cuts on the O-rings, or misshapen or dented housings.
Pall recommends performing a leak test using the Palltronic® Flowstar® IV, as follows:
- The leak test can be performed on a filtration system with or without a filter installed. If a filter is installed, as is the case with capsule filters, the filter must be dry. Diffusive or bulk gas flow through a wet filter cannot be differentiated from leaks in the assembly.
- The outlet of the housing should be sealed with either a blank endcap, valve, or other suitable termination method.
- All drain and vent valves must be in the closed position, except in cases where the connection to the instrument is made through a vent.
- A length of pneumatic tubing with the Palltronic Flowstar IV external vent valve installed should be used to connect the OUT port of the Palltronic® Flowstar IV integrity test instrument to the housing vent port.
- From the Palltronic Flowstar IV main menu, select the “Leak Test” function.
- The Forward Flow test pressure that is used to test the filters should be used as the pressure for the leak test.
- The maximum system size for the leak test is 50 liters.
If the Palltronic Flowstar IV integrity test instrument is not able to detect a pressure loss in the volume or the leak rate is too small to be detected, it will report “no leak detectable” or “flow within limits’ as the result. These results confirm that the filter system under test is free of significant upstream leaks.
If the test reports “flow outside limits” (> 1 mL/min)
- Ensure that the test is being carried out under stable temperature conditions.
- Re-check the connections and housing enclosure are tightened fully.
- If sanitary fittings are an integral part of the filter housing, then the elastomeric seals should be examined and changed if necessary.
- For smaller housings, submerging the pressurized system in a water bath and looking for bubbles will indicate the location of the leak.
- A soap-based leak detecting product can be used around the fittings and connections to locate a leak.
For more information, please contact Pall’s NEW Equipment Support Hotline.
The Bubble Point test measures the pressure region at which diffusive flow transitions into bulk flow, by looking for a deviation from the stable background flow versus flow through open pores. However, a marginal leak in the upstream side of the system under test, or a minor damage to the filter, can add a small amount of gas flow. This could be interpreted by the test instrument as part of the background flow, and therefore, the leak would not be detected. The module factor has been designed into the software of the Palltronic® Flowstar IV software to detect these types of flow contributions.
As stated in USD 2594, Instructions for use for the Palltronic® Flowstar IV integrity test instrument, the Module Factor (MF) defines the sensitivity for the Leak Test phase which is executed at the beginning of the Bubble Point test sequence.
The Bubble Point integrity test using the Flowstar IV integrity test instrument features this stabilization/leak test before proceeding to the actual step-wise increase of pressure for the “Bubble Point” test. This initial leak test seeks to establish that filter system under test shows an expected and typical background flow.
For this test, the upstream side of the filter is pressurized to 80% of the minimum programmed BP value, followed by a measurement of the gas flow occurring at this gas pressure, which is compared against a limit value represented by the programmed filter type related ‘Module Factor’. If the gas flow measured is below the limit value, the BP test proceeds.
The lower the “Module factor” is set, the more sensitive it will be to detect a deviation from the typical background flow for the filter under test during this step in the test sequence.
The expected, typical and thus acceptable background flow caused by gas diffusion is dependent on the filter area. Pall recommends that the “Module Factor” should be set to the number of 10” modules being tested. It should be set to 1.0 for 10” cartridges, 0.5 for 5” cartridges (e.g. AB05 or SLK7002), 0.2 for smaller (e.g. AB02 or SLK7001) filter and 0.1 for flat membranes (e.g. 142 mm discs) or mini capsules (KA02 or similar). The default value is 1 if nothing is entered.
This will ensure that the test sequence is aborted when a filter clearly displays a gas flow above the expected typical background value.
For software versions > 2.0b, the user can set the Module Factor to as low as 0.01 for small area filters such as the Acro 25/50 and KM5 devices. See Table I below for the recommended input values for the module factor.
For more information, please contact Pall’s NEW Equipment Support Hotline.
The Bubble Point specification provided on the Certificate of Test is a manufacturing specification for the filter membrane and should not be used by the end-user as a test limit for the final device. The Manufacturing specifications limit value relies on a defined endpoint and is therefore has a defined end-point. This uses a different Bubble Point test method than what the end-user performs (which inherently is more subjective).
Pall provides a certificate of test with each sterilizing grade filter. The first paragraph Certificate of Test discusses the membrane Bubble Point. An example of this statement is as follows: The 0.2 µm filter membrane used in the filter element has a quantitative bubble point (i.e. "KL") which met or exceeded 3655 mbar (53.0 psi) in water.
As stated, this value is the minimum specification of the flat sheet membrane that is used in the device, not the specification of the device itself. This test, termed the Quantitative Bubble Point (QBP), is objectively and quantitatively defined as the pressure needed to achieve a specified air flow limit through a specified membrane area. This test is conducted on samples from every sterilizing grade membrane roll produced by Pall and reflects the largest pores in each entire roll, to be used either in discs or in cartridge production.
The minimum expected Bubble Point (BP) limit provided for cartridge filters are typically lower than the minimum membrane QBP. The lower end-user Bubble Point limit considers the lower observed Bubble Point typically seen with increasing filter area. It also considers the variability in BP measurements due different algorithms used by different test instruments, as well as variability due to human interpretation of the test result when a visual (manual) Bubble Point test is used.
Please contact Pall Corporation if you require minimum Bubble Point test parameters.